The group label for carrier disinfectants has been released. Disinfectant wipes How will the formula be developed in the future?
This National Day holiday is a great time for friends who care about infection control and disinfection industries. Run. In addition to the grand celebration of the 70th anniversary of the founding of the People’s Republic of China, the majority of colleagues who have been looking forward to the standard for disinfectant wipes have finally ushered in the release of the standard for carrier disinfectants. Although it was downgraded to a group label after coordination between all parties, disinfectant wipes were finally given a formal status, and its significance is self-evident.
Disinfectant wipes are still in their infancy in China, and their development in categories, uses, efficacy and other aspects is still far behind foreign countries. This article will interpret for you the index requirements of the carrier disinfectant group label for disinfectant wipes, raise doubts about the detection method, draw on the development experience of the foreign disinfectant wipes industry, and express my own opinions on the future direction of the disinfectant wipes formula.
Follow this public account and reply to “Carrier Disinfectant” to obtain the PDF version of the full text of hygienic requirements and preparation instructions for carrier disinfectants.
Statement: This article’s interpretation of the carrier disinfectant group label represents only my personal opinion. It does not represent the will of national regulatory agencies, nor does it represent the interests of any company. Please forgive me if there is any misreading.
Form of carrier disinfectant: disinfectant (liquid, solid) + carrier (cloth, fabric, cotton, sponge). According to the carrier, it can be divided into disinfectant wipes, disinfectant dry wipes, disinfectant sponges, and disinfectant cotton balls (sticks). Usage objects: hygienic hands, skin, mucous membranes, environmental surfaces, ordinary object surfaces, medium and low-risk medical device surfaces, medical supplies surfaces, and items and environments contaminated by dirt. Compared with the previous use objects of liquid disinfectants, it does not include: high-level disinfection and sterilization of medical instruments and endoscopes, disinfection of surgical hands, water bodies, disinfection of fabrics and porous object surfaces, disinfection of eating and drinking utensils, food equipment, and fruits. Air disinfection.
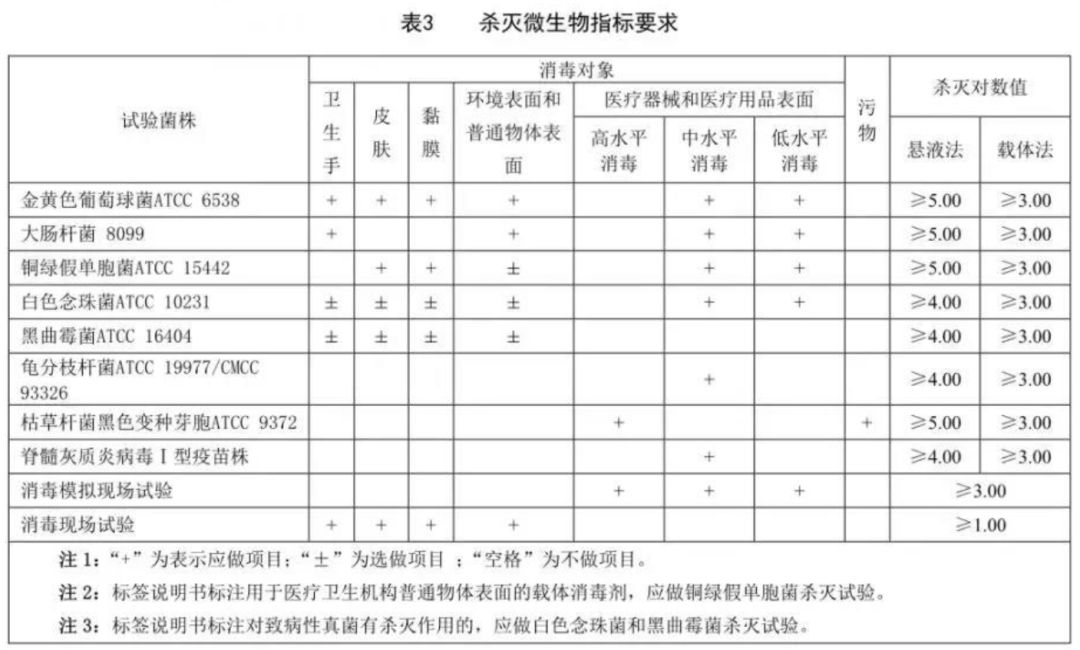
As for the technical content in the carrier disinfectant group label, a simple sentence is as follows:
T/WSJD 001- Hygienic requirements for carrier disinfectants in 2019 ≈ In accordance with the testing items in WS 628-2018 “Technical Requirements for Hygiene and Safety Evaluation of Disinfection Products” (hereinafter referred to as “Hygienic Evaluation Requirements”) and the 2002 edition of “Technical Specifications for Disinfection” (hereinafter referred to as “Technical Specifications for Disinfection”) ) testing method to operate the enhanced version of WS 575-2017 “Hygienic Requirements for Sanitary Wipes” (hereinafter referred to as “Sanitary Wipes Requirements”).
Now let’s focus on the part where “≈” has been omitted:
Part 5 raw material requirements, requirements for initial contaminating bacteria on the carrier ≤ 200cfu/g, requirements for sanitary wipes Not mentioned in . The content of lead, arsenic, and mercury in disinfectants shall not exceed 10 mg/kg, 2 mg/kg, and 1 mg/kg respectively. The values in the sanitary wipes requirements are 40, 10, and 1.
Part 6 Technical Requirements, The liquid content ratio is 1.7~5.0, and the upper limit of 5.0 is not mentioned in the sanitary wipes requirements. Of course, this upper limit cannot be reached by ordinary wet wipes products. Disinfectant dry wipes, wood pulp composite cloth disinfectant wipes and disinfectant sponges are not subject to this restriction. The pH value of the carrier disinfectant is required to be within ±1 of the labeled value. Sanitary wipes require that the pH value of the extruded liquid of sanitary wipes used on hands, skin, and mucous membranes be 3.5 to 8.5, and the pH requirements for other types are not mentioned.
For use on the surface of medical devices and metal objects, the production fluid is required to be basically non-corrosive to stainless steel and non-corrosive or slightly corrosive to carbon steel, aluminum and copper. Compared with sanitary wipes, the metal should be basically non-corrosive and slightly relaxed. Since the concentration of disinfectant in disinfectant wipes is higher than that of ordinary wipes, it is almost impossible to achieve no corrosion on carbon steel, aluminum, and copper. It is also not easy to achieve mild corrosion on aluminum and copper. In some high concentration formulations it may be necessary to add metal corrosion inhibitors. Commonly used inorganic salt corrosion inhibitors, such as sodium molybdate, may reduce the solubility of cations such as quaternary ammonium salts and cause stratified precipitation. Therefore, it is necessary to pay attention to the stability of the formula. Personally, I think this rule is a bit strict. After all, materials such as aluminum and copper rarely appear in hospital environments, and even moderate corrosion does not affect their use. The standard also says that if it is used on specific instruments or metal surfaces, it only needs to be non-corrosive or slightly corrosive to the corresponding material. But do environmental common surface wipes count as specific metal surfaces? It doesn’t seem to count. Let’s see the Ministry of Health’s interpretation of this later. Of course, the level of carrier disinfectant is a group standard, not a national mandatory standard. If the manufacturer feels that it is not suitable for its own product, it does not need to be referenced, as long as the client can explain it.
In terms of microbial indicators, those used on damaged skin and mucous membranes require sterility. Other products require the total number of bacterial and fungal colonies to be ≤10cfu/g, and pathogenic bacteria must not be detected. Compared with sanitary wipes, the requirements have been strengthened. For the testing items of microbial killing indicators, the corresponding strains should be selected according to the purpose for laboratory sterilization experiments. The scope of selection includes: the killing log number of P. aeruginosa suspension method is ≥ 5, the killing rate of koji albicans is ≥ 4, and the killing rate of Mycobacterium chelonae and poliovirus suspension method is ≥ 4 for medium-level disinfection. The requirements in this part are the same as the disinfection technical specifications. In addition, disinfection simulation field experiments are required for medical devices and medical supplies, and disinfection field experiments are required for disinfection of hands, skin, mucous membranes, environmental surfaces and ordinary object surfaces. In the hygiene evaluation requirements, the requirements for hands, skin, mucous membranes and object surfaces are for on-site practice.�The action time is enough. This can also be regarded as a complement to the previous detection of sterilization rate of extruded liquid. If the production fluid is also used for testing in on-site experiments, the sterilization effect data produced will have no reference value.
Okay, the discussion on the content of the standards ends here. Although there are still some details that require further clarification and explanation by relevant departments and agencies, the overall guidelines for disinfectant wipes are very clear and the indicator requirements are clarified. and detection methods. Let’s take a look at some of the problems with current mainstream disinfectant wipes formulas.
Take a well-known British disinfectant wipes brand as an example. Its main ingredient is 0.8% to 0.9% quaternary ammonium salt, compounded with a small amount of other sterilizing and antiseptic ingredients. Its active ingredient content far exceeds the 0.3% to 0.4% quaternary ammonium salt added by its domestic counterparts. The main reason for this is that in foreign countries, intermediate-level disinfection can be claimed by killing a portion of enveloped viruses and mycobacteria. However, in China, due to different regulations, medium-level disinfection requires killing poliovirus, which cannot be achieved with quaternary ammonium salts. Therefore, under the domestic regulatory framework, such a high addition amount cannot bring significant differences in product claims. (There are still differences in market guidance.) At the same time, the disadvantages of higher addition amounts are also obvious. There are many market feedbacks: pungent smell; skin irritation; metal corrosion; slippery feel; foam; obvious water stains. To be honest, the sterilization effect is indeed good, but there are still many other aspects that can be improved.
In addition to the cationic system, other common ones include 0.63% sodium hypochlorite, alcohol/isopropyl alcohol 50%~70% compounded with quaternary ammonium salts or chlorhexidine. These are relatively traditional disinfectant wipes formula systems. Diversey’s 0.5% hydrogen peroxide system can kill mycobacteria and polio, achieve a medium-level disinfectant effect, and is non-irritating to the skin and eyes, with no disinfectant residue and no obvious corrosiveness. It can be said to be the highlight of the disinfectant wipes formula in recent years. In terms of disinfection dry wipes, the main technical route is to react with TAED (tetraacetylethylenediamine) and a hydrogen peroxide source (such as sodium percarbonate) in contact with water to generate peracetic acid. The following recipe is sourced from the Lubrizol Corporation website.
In recent years, foreign countries, especially the European Union, have increasingly stricter controls on sterilization and disinfection products, and regulatory thresholds are getting higher and higher. Foreign disinfectant manufacturers pay more attention to regulatory registration barriers and investment, but have little to say about improvements in formula technology. Since the regulatory registration cost for disinfectant products in Europe is as high as millions, many foreign disinfectant brands still use formulas from ten years ago or even twenty or thirty years ago. As mentioned earlier, a certain British brand of disinfectant wipes has various shortcomings. These shortcomings also exist in many European and American disinfectant wipes products. However, in order to avoid the huge cost of re-registering the formula, these defective formulas are still used without any improvement for 20 to 30 years.
Looking back at the domestic disinfectant wipes industry, although the domestic terminal market has only just emerged in recent years. However, as China is a major exporter of non-woven fabrics and a major manufacturer of wet wipes, some wet wipes factories have long joined the international industrial chain of disinfectant wipes. After years of accumulation of technical experience, some manufacturers no longer just manufacture products, but also start exporting formulas. The formula of disinfectant wipes produced by some engineers is superior to that of foreign brands in terms of sterilization effect and other sensory indicators. In addition, domestic regulations on disinfection products are still relatively loose, and product registration fees are still within the acceptable range of most companies. In the future, domestic disinfectant wipes have great potential in product innovation, and it is completely foreseeable that they will surpass international brands in product performance and quality!






